Friday, 14 February
In class
- Present the equilibrium case studies.
Assignments
- On Tuesday we will begin our study of acid-base equilibria. Begin reading Chapter 14 in your textbook and working as many of the exercises as time allows over the next two weeks.
- The equilibrium exam will be given on Friday, 21 February.

Thursday, 13 February
In class
- Ask any questions about the experiment analysis. Finish this to turn in tomorrow.
- Finish your group work on the case study and practice your presentation for tomorrow.
Assignments
- Molar Solubility of Calcium Hydroxide paper due tomorrow.
- Finish any last preparations for the case study presentations tomorrow.
Wednesday, 12 February
In class
- Review the pre-experiment questions and ask any questions you may have.
- Collect data on the solubility of calcium hydroxide.
Assignments
- Plot your data and find the best two linear regression lines to determine the maximum mass of calcium hydroxide that dissolves in 500 mL of water.
- Answer the analysis questions.
- Finish the equilibrium unit progress check exercises.
Tuesday, 11 February
In class
- Observe the relationship between solubility and conductivity.
- Read and complete the pre-experiment sections for the Solubility of Calcium Hydroxide experiment.
- Continue work on the equilibrium case study with your group.
Assignments
- Watch Effects of Concentration on Conductivity Using Go Direct™ Conductivity to learn how to use the conductivity probe for tomorrow’s experiment.
- Continue work on the equilibrium unit Progress Check
Monday, 10 February
In class
- Learn to spot another common simplification for equilibrium calculations.
- Form working groups and choose your unit case study. (Case studies uploaded to the secure page.)
Assignments
- Begin work on the Equilibrium Unit Progress Check in APC.
Friday, 7 February
In class
- Take the quiz on equilibrium calculations.
Thursday, 6 February
In class
- Whiteboard presentations: equilibrium calculations using the ICE table method.
- Review and discuss your performance on the thermochemistry unit exam.
Assignments
- Review for a quiz on writing equilibrium expressions and calculations.
Wednesday, 5 February
In class
- Show me your attempt at exercises 36 and 40 in Chapter 13 and discuss.
- Explain the effect of diluting an aqueous system at equilibrium.
- Learn how to to calculate the amount of a reactant or product at equilibrium.
Assignments
- Answer exercises 52, 53, 55, and 65 in Chapter 13 before class tomorrow.
Tuesday, 4 February
In class
- Learn how to change the equilibrium position of a chemical system.
Assignments
- Answer exercises 36 and 40 from Chapter 13 in your textbook before the start of class tomorrow.
Monday, 3 February
In class
- Answer today’s Wake Up exercise: Electronegativity.
- Explain the difference between homogeneous and heterogeneous equilibrium.
- Chapter 13 Exercises: 15 and 16.
Assignments
- Dress for laboratory work tomorrow.
Friday, 31 January
In class
- Take the thermochemistry unit exam.
Thursday, 30 January
In class
- Learn how to use the reaction quotient, Q, to determine if a chemical system has reached equilibrium.
Assignments
- Finish your practice and review for the thermochemistry exam tomorrow.
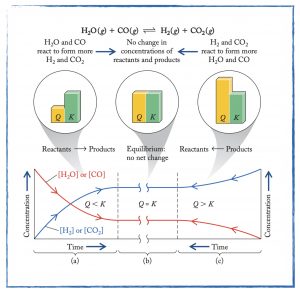
Wednesday, 29 January
In class
- Review for the thermochemistry unit exam to be taken on Friday.
Tuesday, 28 January
In class
- Show me your attempts at the equilibrium exercises in Chapter 13 (1 – 5, 7, 8.).
- Learn to write rate laws for mechanisms that have a reversible elementary step.
Assignments
- Finish work on the thermochemistry Progress Check before the start of class tomorrow.
Monday, 27 January
In class
- Answer today’s Wake Up: Dynamic Equilibrium.
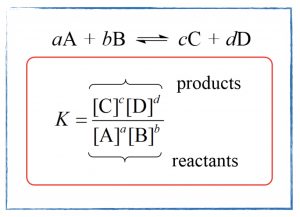
- Analyze equilibrium reaction data and learn to write the equilibrium expression and calculate the equilibrium constant.
- Complete and present the Using Equilibrium Expressions whiteboard problem set exercises.
- Ask any questions on the APC Progress Check questions for thermochemistry.
Assignments
- Complete Exercises 1 – 5, 7, and 8 in Chapter 13.
- Continue your review for the thermochemistry exam on Friday.
Friday, 24 January
In class
- Carryout and analyze a physical analogy of dynamic equilibrium.
- Watch Bailing Beakers if you missed class today or want a review of the analogy.
- Use some simulations to see how reversible reactions and equilibrium work with a chemical reaction.
- PhET simulation: Reversible Reactions
- PhET simulation: Reactions and Rates
Assignments
- Chapter 13: Fundamental Equilibrium Concepts, covers all of the material for this unit. Begin reading and working as many of the end of chapter exercises as time allows.
- Continue your review work for the thermochemistry unit exam next Friday.