Thursday, 23 January
In class
- Answer the Heats of Formation Wake Up.
- Discuss your answers to Part 3 of the Bond Enthalpies problem set.
- Finish review of the fall term final exam.
- Decide upon a day to take the thermochemistry unit exam.
Assignments
- I’ve assigned the Thermochemistry Unit Progress Check in APC. Work the MCQ section before class tomorrow and come with specific questions as necessary.
Wednesday, 22 January
In class
- Answer the Bond Enthalpies Wake Up exercise.
- Review your performance on the final exam and ask any questions.
Assignments
- Finish your work on the Part 3 calculations of the Bond Enthalpies problem set.
Tuesday, 21 January (Start of the Spring Term)
In class
- Share your thoughts and comments about the final exam.
- Learned how to use average bond enthalpy values to calculate the energy change in a chemical reaction.
- Bond Enthalpies problem set.
Assignments
- Research and calculate a value for Part 3 in the Bond Enthalpies problem set by the start of class on Thursday.
Friday, 10 January
In class
- Conduct your calorimetry experiment and submit the paper.
Assignments
- Rest, review, and relax to be ready for your final exams next week. Best of luck.

Thursday, 9 January
In class
- Finish your one-page summary and practice questions and give to me. I’ll scan them and upload a final draft here by the end of the day.
- 3 Green One-Page Summaries
- 4 Green One-Page Summaries
Wednesday, 8 January
In class
- Ask any questions about the experimental set up for your lab practical on Friday. (Equipment, set up, calculations, etc)
- Complete your hand-written/drawn one-page summary of your group’s assigned unit and submit (if possible) so I can combine and upload a copy for each of you.
Assignments
- Keep reviewing for the fall term final exam.
Tuesday, 7 January
In class
- Ask any questions about exercises 64 – 66 in Chapter 9.
- Form your lab working group for the final exam practical on Friday and ask any questions regarding the logistics.
- In your groups, create a one-page summary for your assigned unit.
- One-Page Summaries
- Unit 1 Learning Objectives (Since I didn’t start this until Unit 2.)
- Unit 2 Learning Objectives
Assignments
- Begin reviewing for the final exam. I suggest using the Barron’s AP Chemistry book.
- Bring your Barron’s prep book with you to class tomorrow to aid in your one-page summaries.

Monday, 6 January 2025
In class
- Welcome back and Happy New Year!
- Let me know your preferences about MCQ exam questions for the Spring Term.
- Use Hess’s law to determine the enthalpy of reactions that may be difficult to measure in the laboratory.
- Hess’s Law problem set and group whiteboards.
Assignments
- Complete exercises 64 – 66 in Chapter 9.
- Read section 9.4 up to the middle of p. 459.
Friday, 20 December
In class
- Show me your attempts on the textbook exercises and ask any questions that you still have.
Assignments
- Enjoy your winter break! See you in 2025.

Thursday, 19 December
In class
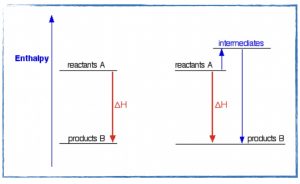
- Investigate standard heats of formation and use them to calculate the energy change for any chemical reaction.
- Discuss and review your work on the kinetics unit exam.
Assignments
- In your notebook work out an answer for Exercises 61, 67, and 68 in Chapter 9 before the start of class tomorrow.
Wednesday, 18 December
In class
- Write an equation that represents the first law of thermodynamics and the relationship between energy, heat, and work.
- Practice writing and calculating with thermochemical equations.
- Thermochemical Equations problem set.
Assignments
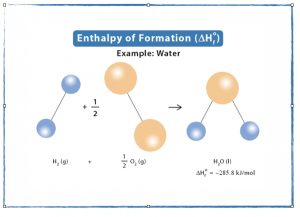
- Read pages 445 – 449 in your textbook discussing standard enthalpies of formation.
Tuesday, 17 December
In class
- Take the kinetics unit exam with your partner.
Assignments
- Finish reading sections 9.1 and 9.2 in the textbook.
- Begin reading section 9.3, p. 441 – 445, stopping when you get to the section titled Standard Enthalpy of Combustion.

Monday, 16 December
In class
- Ask any questions on the calculations for the heat of neutralization experiment.
- Calculate the amount of energy needed to convert a 5.0 kg sample of ice at -5 C to steam at 120 C.
Assignments
- Make yourself ready for the kinetics exam in class tomorrow.
- Printed copy of your CER paper due on Friday.

Friday, 13 December
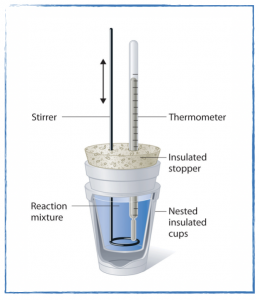
In class
- Set up your experiment and collect temperature change data to determine the enthalpy of neutralization. You will want to run at least three trials to get good data, but I recommend carrying out the calculations after the first trial and comparing your enthalpy change (measured in kJ/mol) to the accepted value.
- Begin writing a CER paper for this experiment and your results. This will be due at some point towards the end of next week.
Assignments
- Continue your review for the kinetics exam on Tuesday next week.
Thursday, 12 December
In class
- Measure the heat of a neutralization reaction by utilizing the technique of calorimetry.
- Answer the pre experiment questions.
- Study the experimental set up and procedure.
- Begin data collection if time allows.
Assignments
- Begin reading Chapter 9 in your textbook.
- Continue to review for the kinetics exam on Tuesday next week.
Wednesday, 11 December
In class
- Take the exam day survey if you didn’t do so yesterday. I need a complete data set! 😉
- Ask any questions you have on kinetics before the exam on Tuesday, 17 December.
- Review the basic concepts and terminology of thermochemistry.
- Energy Forms and Changes PhET simulation.

