Friday, 2 May
Friday, 25 April
In class
- Please complete the following survey regarding our field trip to Sara Lee Bakery Labs on Tuesday, 20 May.
- Diagnostic Test 1 Analysis paper
Thursday, 24 April
In class
- Click here for the scoring guidelines to the 2024 FRQ questions.
- I’ve released 3 practice exams in APC for you to try if you so choose to.
Wednesday, 16 April
In class
- Take the Thermodynamics and Electrochemistry unit exam.

Tuesday, 15 April
In class
- Review for the Thermodynamics and Electrochemistry exam tomorrow.
Monday, 14 April
In class
- Electroplate a washer, key, or other metal item.
- Choose a partner for the Thermodynamics and Electrochemistry exam on Wednesday.
- Feedback on the Acids and Bases retake exam.
Assignments
- Work on the Unit 9 Progress Check in APC.
- Complete the assigned practice quizzes in K.A. before tomorrow morning at 8:00:
- Application of Thermodynamics: Quiz 2
- Applications of Thermodynamics: Unit test
Friday, 11 April
In class
- Discuss electroplating and Faraday’s Law of Electrolysis.
- Answer the pre-experiment questions for the Electroplating with Copper experiment.
Assignments
- Complete Chapter 16 Exercises 43, 47, and 49 by the start of class on Monday.
- If wanted, bring a metallic item to electroplate on Monday.

Tuesday, 8 April
In class
- Use qualitative and quasi-quantitative reasoning to describe the change in voltage of a galvanic cell that is not under standard conditions.
- Complete and present whiteboards on the electrochemistry problem set.
Assignments
- Finish the K.A. lessons before class on Friday.
- Watch Easy Step-by-Step Tutorial on Electroplating.
Monday, 7 April
In class
- Learn why a battery goes dead by calculating the cell potential and the free energy of the galvanic cell reaction.
Assignments
- Complete by Friday the assigned lessons in K.A.
Friday, 4 April
In class
- Take the Acids and Bases unit retake exam.
- Watch Voltaic cell | How does it work?
- Use the half-cell plate and voltage meter to measure the voltage of various galvanic cells.
Assignments
- Read sections 16.2 – 16.3 in the text before the start of class on Monday.
- Answer exercises 11 and 21 in Chapter 16.

Thursday, 3 April
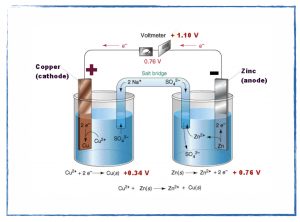
In class
- Discuss the zinc and copper(II) sulfate redox reaction particle diagrams.
- Convert the zinc and copper(II) sulfate reaction into an electrochemical cell.
- Briefly review for the Acids and Bases retake exam tomorrow.
Assignments
- Review for the retake exam.
Wednesday, 2 April
In class
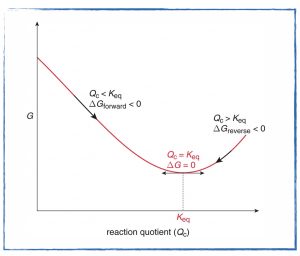
- Check in and discuss the Gibbs Energy and Equilibrium problem set.
- Take the Free Energy and Equilibrium (practice) quiz in APC and discuss.
- Listen to an introductory lecture on electrochemistry and the quest for cleaner fuel and review simple redox reactions.
Assignments
- Watch the KA video Coupled Reactions.
- Take the Applications of Thermodynamics: Quiz 1 in KA before the start of class on Friday.
Tuesday, 1 April
In class
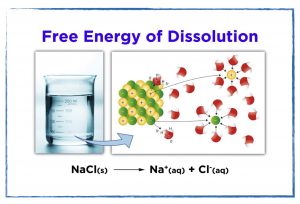
- Create particle diagrams for the process of dissolution to help explain the changes in enthalpy and entropy.
- Describe the difference between Gibbs free energy and standard Gibbs free energy.
- Work on the Gibbs Energy and Equilibrium problem set.
Assignments
- Complete the Gibbs Energy and Equilibrium problem set before class tomorrow.
- Continue reviewing Part 1 in Barron’s.
- Look over exercises for the Acids and Bases retake on Friday.
Monday, 31 March
In class
- Answer the wake up exercise on thermodynamic and kinetic stability.
- Watch M&Ms Dissolving in Water
- Explain the driving forces for the dissolution of sodium chloride versus magnesium chloride.
Assignments
- Complete the KA assignment Free Energy and Equilibrium before the start of class tomorrow.
- It’s time to begin reviewing for the College Board exam! Start this week with Part 1: Structure of Matter in the Barron’s book. Aim to finish the two chapters of this section by Friday.
Friday, 21 March
In class
- Answer the wake up question on free energy.
- Review your performance on the Acids and Bases unit exam.
Thursday, 20 March
In class
- Calculate the change in entropy of the universe for:
- ice melting and
- nitrogen and hydrogen gas forming ammonia.
- Develop a good working definition of free energy.
- With your assigned group, answer the questions in the Free Energy problem set.
Assignments
- Finish work on the problem set before class tomorrow. Your assigned groups will present a whiteboard of selected questions the Monday we return from Spring Break.
Wednesday, 19 March
In class
- Answer a bonus question on creating a buffer solution.
Assignments
- Read section 12.3 in your textbook on the second law of thermodynamics.
Tuesday, 18 March
In class
- Take the Acids and Bases unit exam.

Monday, 17 March
In class
- Ask questions and review for the Acids and Bases unit exam tomorrow.
Friday, 14 March
In class
- Answer and present the entropy whiteboard problem set.
- Q&A on acid/base topics for the unit exam next Tuesday.
Assignments
- Complete the Gibbs free energy and thermodynamic favorability lesson in Khan Academy.
- Continue reviewing for the Acids and Bases unit exam on Tuesday.

Thursday, 13 March
In class
- Set up your Khan Academy account for this unit.
- Classify a set of processes as either spontaneous or non spontaneous.
- Ask questions about topics for the test on Tuesday.
Assignments
- Complete the assigned lesson, Entropy, in Kahn Academy by class tomorrow.


