Wednesday, 12 March
In class
- Prepare and test your buffer solutions. Two attempts remaining.
- Take the Acids and Bases Diagnostic Quiz.
Assignments
- Continue reviewing for the Acids and Bases Unit Exam next on Tuesday.
Tuesday, 11 March
In class
- Prepare and test your buffer solutions. You have a total of three attempts.
Assignments
- Finalize one copy per group of the buffer experiment and submit.
- Continue reviewing for the Acids and Bases Unit Exam, next Tuesday.
Monday, 10 March
In class
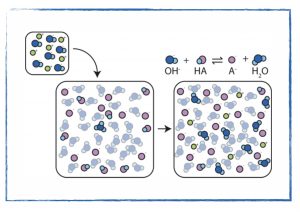
- Visualize a buffer solution at the molecular level.
- Calculate the change in pH when strong acid is added to a) pure water and b) buffered river water.
- Design and prepare a buffer for the study of a biochemical reaction.
Assignments
- Dress for laboratory work tomorrow.
- Begin work on the Unit 8 Progress Check.
- Take the survey on your preferred day for the Acid-Base Unit Exam.
Friday, 7 March
In class
- Determine if the dissolved salt, sodium sulfite, will create an acidic, neutral, or basic solution.
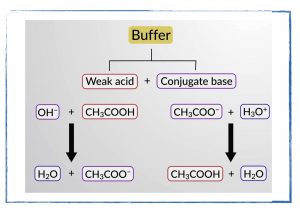
- Derive an equation that will allow us to easily determine the ph of solutions that are a mixture of acids and bases.
Assignments
- Read section 14.6 in your textbook on buffers.
Thursday, 6 March
In class
- Calculate the pH of rain water and acid rain in which there are acids that can donate more than one hydrogen ion.
- Complete the Acid and Base pH Calculations whiteboard set and present your findings to the class.
Assignments
- Watch this on calculating the pH of salt solutions.
- Read p. 414 – 417 in the Barron’s Review book for more explanation and examples of the pH of salt solutions.

Wednesday, 5 March
In class
- Determine the base dissociation constant value for the weak base, trimethylamine.
Assignments
- Catch up with the problem sets as necessary.
Tuesday, 4 March
In class
- Catch up on what you worked through during my absence last Friday.
- Discuss feedback on the equilibrium unit exam.
- Take the solubility equilibrium retake question.
- Finish the Comparing Titration Curves problem set.
Assignments
- Complete Model 1 (questions 1 – 20) of the Weak Acid / Strong Base Titration pH Calculations problem set before the start of class tomorrow.
Friday, 28 February
In class
- Begin work on the Weak Acid / Strong Base Titration pH Calculations problem set.
Thursday, 27 February
In class
- Discuss the APC practice quizzes.
- At this point we have covered most of the key concepts for acids and bases. The next few days will be spent practicing with this material in order to become more comfortable. Complete the following two problem sets:
- Titration Curves
- Comparing Titration Curves
Assignments
- By now you should have read through 14.3 in your textbook. Continue on now with sections 14.4 – 14.6. Complete as many of the exercises at the end of the chapter as time allows and/or Chapter 13 of the Barron’s Review book.
- As requested in Block 3 today:
Wednesday, 26 February
In class
- Discuss and compare your titration plots to the standard plots.
- Practice quizzes (APC) on pH.
Assignments
- Complete the Weak Acid and Base Equilibria quiz in APC before the start of class tomorrow.
Tuesday, 25 February
In class
- Check in and discuss Chapter 14 exercises 19, 40, and 42.
- Plot pH data for the titration of samples of hydrochloric acid and acetic acid.
Assignments
- Plot your data from the titrations experiments: volume of NaOH added (mL) on the x axis and pH on the y axis. Connect your points with a smooth line.
Monday, 24 February
In class
- Compare the pH of a 0.1 M HCl solution to a 0.1 M HF solution.
Assignments
- Read section 14.3 in your textbook.
- Completed Chapter 14 exercises 19, 40, and 42 before class tomorrow.
Friday, 21 February
In class
- Take the equilibrium unit exam.
Thursday, 20 February
In class
- Turn in your group’s data table.
- Explain why there is a difference in measured pH between 0.1 M hydrochloric acid and 0.1 M acetic acid solutions.
- Ask any last questions before the equilibrium exam tomorrow.
Assignments
- Bring a charged device with the lockdown browser installed for the multiple choice section of the exam tomorrow.
Wednesday, 19 February
In class
- Review and discuss the common-ion effect question from APC.
- Use our probes to measure the pH of a number of common acids and bases and calculate the concentration of hydronium ions in each sample.
Assignments
- Finish the data table and print one copy to turn in for your group.
Tuesday, 18 February
In class
- Take the practice quiz in APC on a few applications of chemical equilibrium.
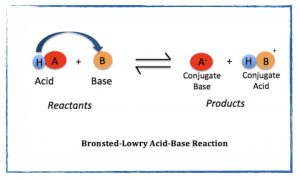
- Identify the Bronsted-Lowry acid, base, conjugate acid, and conjugate base in the auto ionization of water reaction.
Assignments
- Dress for laboratory work tomorrow.
- Read through the end of section 14.2 in your textbook before class tomorrow.