Tuesday, 10 December
In class
- Discuss the Catalysis Quiz practice questions from APC.
- Choose your partner for the Unit 5 exam on Friday.
- Review topics for the exam with your partner using chemquiz.net.
Assignments
- Come to class tomorrow with any questions on the Unit 5 topics.
Monday, 9 December
In class
- Answer any questions from the Kinetics Practice Quiz in APC you worked on last Friday.
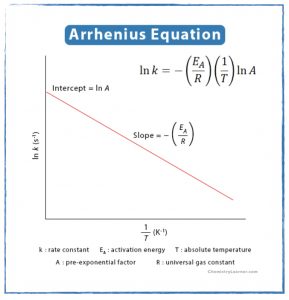
- Determine the relationship between temperature and a chemical reaction’s rate law.
- Why is chlorine an efficient ozone destroyer?
Assignments
- Play around with ChemQuiz.net and see if you find it useful.
- Try the Catalysis Quiz practice questions in APC.
- Begin thinking about whom you would like to work with for the Unit 5 exam on Friday.
Friday, 6 December
In class
- Work through the Kinetics Practice Quiz in APC and make note of any concepts that are still not clear to you.
Thursday, 5 December
In class
- Take the practice quiz on the graphical determination of integrated rate laws.
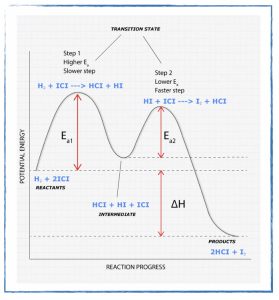
- Explain what a reaction mechanism is and the different ways chemists attempt to determine them for a given chemical reaction.
Assignments
- Finish the Reaction Mechanisms and Energy Diagrams problem set if you didn’t do so in class today.
Wednesday, 4 December
In class
- Discuss your work on the reactions rate quiz.
- Ask any questions about the method of initial rates exercises assigned yesterday.
Assignments
- Another short quiz tomorrow. This one will cover the ideas in the Graphical Determination of Rate Laws problem set.
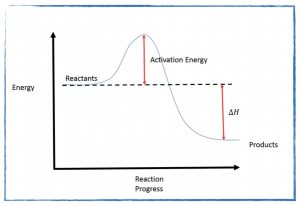
- Read section 17.6 in your textbook on Reaction Mechanisms. This is a challenging topic that we will discuss in detail tomorrow but reading ahead will help you have some familiarity with the concepts.
Tuesday, 3 December
In class
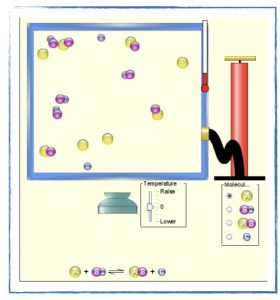
- Take the quiz on reaction rates and collision theory.
- Practice with a concentration v time data set to determine the order of the decomposition of ozone gas.
- What happens if our reactions have more than one reactant?
- Learn how to use the method of initial rates.
Assignments
- Practice some method of initial rate problems: Chapter 17, 25 – 27.
Monday, 2 December
In class
- How can the rate constant of a reaction be determined from rate data?
- Integrated Rate Laws problem set.
Assignments
- Finish the problem set from class today if you didn’t do so during our time together,.
- Review for a brief quiz on reaction rates and collision theory tomorrow.
Tuesday, 26 November
In class
- Show me your work on the Chapter 17 Exercises assigned last week and ask any questions you may have.
- Collect data to determine the approximate rate law for the combustion of wax reaction in a burning candle.

Monday, 25 November
In class
- Review and discuss the questions on the Unit 4 exam.
- Determine the best explanation to Extension Question 14 from the Graphical Determination of Rate Laws problem set.
Assignments
- Finish the exercises assigned on Friday to be checked in at the start of class tomorrow.
Friday, 22 November
How can graphical analysis of a chemical reaction rate allow us to determine reaction orders and half-lives?
In class
- Warm up exercise: Sulfur dioxide reacts with oxygen gas to form sulfur trioxide. If the initial rate of change for sulfur dioxide is -2.00 M/min, what is the initial rate of change of oxygen and sulfur trioxide?
- Explain what a rate law tells us about a chemical reaction and use graphical data to determine the reaction order.
Assignments
- Using the Reactions and Rates PhET simulation, answer Exercises 9 – 11 in Chapter 17 in your textbook.
- Work out answers to Exercises 1, 2, 4, and 32 in Chapter 17 in your text book.
Thursday, 21 November
In class
- Discuss how the collision theory model can be useful in partially explaining the rates of chemical reactions.
- Explain how certain variables affect the rate of a reaction according to the collision theory model.
Assignments
Wednesday, 20 November
In class
- Set up your experiments and collect data.
- Begin analyzing your data and create graph(s).
Assignments
- Begin reading Chapter 17: Kinetics in your textbook. We will cover all the material in this chapter by the end of the unit.

Tuesday, 19 November
In class
- With your group, design the procedure for the reaction speed experiment.
Assignments
- Dress for laboratory work tomorrow.
- Begin work on the Rate of Reaction problem set. This paper will outline the ways chemists measure the speed of chemical reactions.
Monday, 18 November
In class
- Unit 4 exam.

Friday, 15 November
In class
- Prepare your laboratory notebooks for collection on Monday by putting a sticky note in the margins by the start of the Eight Chemical Reactions experiment so I can flip to it quickly.
- Form lab groups and discuss the central challenge of the How long will that marble statue last? experiment. Then, work your way through the rest of p. 83 and 84 brainstorming your ideas.
- Ask any final questions you have about the material for Monday’s test.
Outside of class
- Review and prepare for the Unit 4 exam on Monday.


